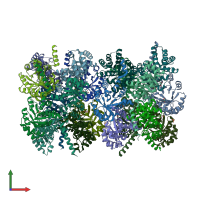
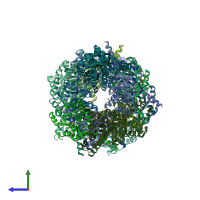
Function and Biology Details
Reaction catalysed:
Sedoheptulose 7-phosphate + D-glyceraldehyde 3-phosphate = D-erythrose 4-phosphate + D-fructose 6-phosphate
Biochemical function:
Biological process:
Cellular component:
Structure analysis Details
Assembly composition:
homo decamer (preferred)
Assembly name:
Transaldolase (preferred)
PDBe Complex ID:
PDB-CPX-194505 (preferred)
Entry contents:
1 distinct polypeptide molecule
Macromolecule: