Function and Biology Details
Reaction catalysed:
S-adenosyl-L-methionine + glycine = S-adenosyl-L-homocysteine + sarcosine
Biochemical function:
Biological process:
Cellular component:
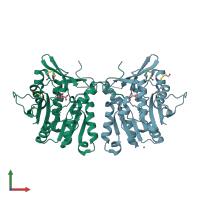
Structure analysis Details
Assembly composition:
homo tetramer (preferred)
Assembly name:
Glycine N-methyltransferase (preferred)
PDBe Complex ID:
PDB-CPX-172093 (preferred)
Entry contents:
1 distinct polypeptide molecule
Macromolecule: