EMD-2664
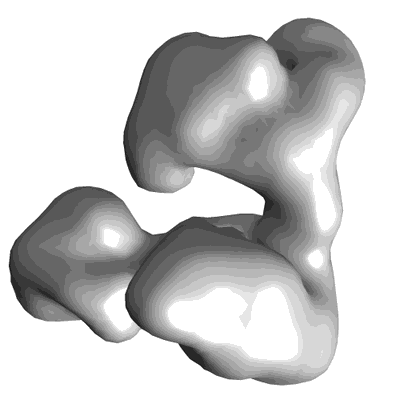
Structure of SMG1C-UPF1 complex, comprising SMG1 kinase, SMG8, SMG9 and UPF1
EMD-2664
Single-particle20.9 Å
 Deposition: 27/05/2014
Deposition: 27/05/2014Map released: 09/07/2014
Last modified: 20/08/2014
Sample Organism:
Homo sapiens
Sample: SMG1C-UPF1 complex, comprising SMG1 kinase, SMG8, SMG9 and UPF1
Deposition Authors: Melero R ,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S
,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S  ,
Yamashita A,
Llorca O
,
Yamashita A,
Llorca O 
Sample: SMG1C-UPF1 complex, comprising SMG1 kinase, SMG8, SMG9 and UPF1
Deposition Authors: Melero R
 ,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S
,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S  ,
Yamashita A,
Llorca O
,
Yamashita A,
Llorca O 
Structures of SMG1-UPFs complexes: SMG1 contributes to regulate UPF2-dependent activation of UPF1 in NMD
Melero R  ,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S
,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S  ,
Yamashita A,
Llorca O
,
Yamashita A,
Llorca O 
(2014) Structure , 22 , 1105 - 1119
 ,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S
,
Uchiyama A,
Castano R,
Kataoka N,
Kurosawa H,
Ohno S  ,
Yamashita A,
Llorca O
,
Yamashita A,
Llorca O 
(2014) Structure , 22 , 1105 - 1119
Abstract:
SMG1, a PI3K-related kinase, plays a critical role in nonsense-mediated mRNA decay (NMD) in mammals. SMG1-mediated phosphorylation of the UPF1 helicase is an essential step during NMD initiation. Both SMG1 and UPF1 are presumably activated by UPF2, but this regulation is incompletely understood. Here we reveal that SMG1C (a complex containing SMG1, SMG8, and SMG9) contributes to regulate NMD by recruiting UPF1 and UPF2 to distinct sites in the vicinity of the kinase domain. UPF2 binds SMG1 in an UPF1-independent manner in vivo, and the SMG1C-UPF2 structure shows UPF2 recognizes the FRB domain, a region that regulates the related mTOR kinase. The molecular architectures of several SMG1C-UPFs complexes, obtained by combining electron microscopy with in vivo and in vitro interaction analyses, competition experiments, and mutations, suggest that UPF2 can be transferred to UPF1 within SMG1C, inducing UPF2-dependent conformational changes required to activate UPF1 within an SMG1C-UPF1-UPF2 complex.
SMG1, a PI3K-related kinase, plays a critical role in nonsense-mediated mRNA decay (NMD) in mammals. SMG1-mediated phosphorylation of the UPF1 helicase is an essential step during NMD initiation. Both SMG1 and UPF1 are presumably activated by UPF2, but this regulation is incompletely understood. Here we reveal that SMG1C (a complex containing SMG1, SMG8, and SMG9) contributes to regulate NMD by recruiting UPF1 and UPF2 to distinct sites in the vicinity of the kinase domain. UPF2 binds SMG1 in an UPF1-independent manner in vivo, and the SMG1C-UPF2 structure shows UPF2 recognizes the FRB domain, a region that regulates the related mTOR kinase. The molecular architectures of several SMG1C-UPFs complexes, obtained by combining electron microscopy with in vivo and in vitro interaction analyses, competition experiments, and mutations, suggest that UPF2 can be transferred to UPF1 within SMG1C, inducing UPF2-dependent conformational changes required to activate UPF1 within an SMG1C-UPF1-UPF2 complex.
