EMD-27947
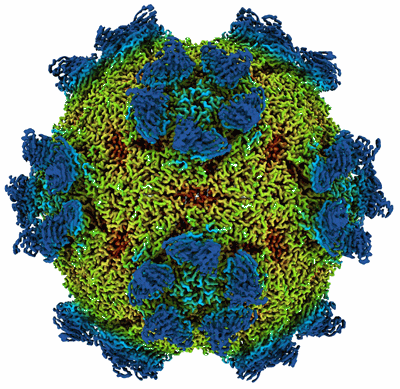
9H2 Fab-Sabin poliovirus 3 complex
EMD-27947
Single-particle2.66 Å
 Deposition: 25/08/2022
Deposition: 25/08/2022Map released: 21/06/2023
Last modified: 16/10/2024
Sample Organism:
Human poliovirus 3 strain Sabin,
Homo sapiens
Sample: Human poliovirus 3 strain Sabin
Fitted models: 8e8r (Avg. Q-score: 0.595)
Deposition Authors: Charnesky AJ
Sample: Human poliovirus 3 strain Sabin
Fitted models: 8e8r (Avg. Q-score: 0.595)
Deposition Authors: Charnesky AJ
A human monoclonal antibody binds within the poliovirus receptor-binding site to neutralize all three serotypes.
Charnesky AJ,
Faust JE,
Lee H  ,
Puligedda RD,
Goetschius DJ
,
Puligedda RD,
Goetschius DJ  ,
DiNunno NM,
Thapa V,
Bator CM,
Cho SHJ,
Wahid R,
Mahmood K,
Dessain S,
Chumakov KM,
Rosenfeld A,
Hafenstein SL
,
DiNunno NM,
Thapa V,
Bator CM,
Cho SHJ,
Wahid R,
Mahmood K,
Dessain S,
Chumakov KM,
Rosenfeld A,
Hafenstein SL 
(2023) Nat Commun , 14 , 6335 - 6335
 ,
Puligedda RD,
Goetschius DJ
,
Puligedda RD,
Goetschius DJ  ,
DiNunno NM,
Thapa V,
Bator CM,
Cho SHJ,
Wahid R,
Mahmood K,
Dessain S,
Chumakov KM,
Rosenfeld A,
Hafenstein SL
,
DiNunno NM,
Thapa V,
Bator CM,
Cho SHJ,
Wahid R,
Mahmood K,
Dessain S,
Chumakov KM,
Rosenfeld A,
Hafenstein SL 
(2023) Nat Commun , 14 , 6335 - 6335
Abstract:
Global eradication of poliovirus remains elusive, and it is critical to develop next generation vaccines and antivirals. In support of this goal, we map the epitope of human monoclonal antibody 9H2 which is able to neutralize the three serotypes of poliovirus. Using cryo-EM we solve the near-atomic structures of 9H2 fragments (Fab) bound to capsids of poliovirus serotypes 1, 2, and 3. The Fab-virus complexes show that Fab interacts with the same binding mode for each serotype and at the same angle of interaction relative to the capsid surface. For each of the Fab-virus complexes, we find that the binding site overlaps with the poliovirus receptor (PVR) binding site and maps across and into a depression in the capsid called the canyon. No conformational changes to the capsid are induced by Fab binding for any complex. Competition binding experiments between 9H2 and PVR reveal that 9H2 impedes receptor binding. Thus, 9H2 outcompetes the receptor to neutralize poliovirus. The ability to neutralize all three serotypes, coupled with the critical importance of the conserved receptor binding site make 9H2 an attractive antiviral candidate for future development.
Global eradication of poliovirus remains elusive, and it is critical to develop next generation vaccines and antivirals. In support of this goal, we map the epitope of human monoclonal antibody 9H2 which is able to neutralize the three serotypes of poliovirus. Using cryo-EM we solve the near-atomic structures of 9H2 fragments (Fab) bound to capsids of poliovirus serotypes 1, 2, and 3. The Fab-virus complexes show that Fab interacts with the same binding mode for each serotype and at the same angle of interaction relative to the capsid surface. For each of the Fab-virus complexes, we find that the binding site overlaps with the poliovirus receptor (PVR) binding site and maps across and into a depression in the capsid called the canyon. No conformational changes to the capsid are induced by Fab binding for any complex. Competition binding experiments between 9H2 and PVR reveal that 9H2 impedes receptor binding. Thus, 9H2 outcompetes the receptor to neutralize poliovirus. The ability to neutralize all three serotypes, coupled with the critical importance of the conserved receptor binding site make 9H2 an attractive antiviral candidate for future development.
