EMD-31252
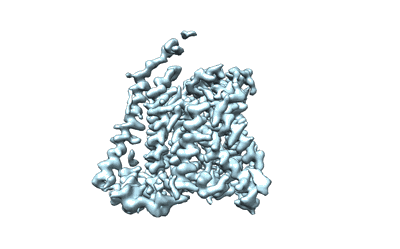
cryo EM map of Erastin-bound xCT-4F2hc complex, focused refined on transmembrane region
EMD-31252
Single-particle3.4 Å
 Deposition: 28/04/2021
Deposition: 28/04/2021Map released: 11/05/2022
Last modified: 11/05/2022
Sample Organism:
Homo sapiens
Sample: cryo EM map of Erastin-bound xCT-4F2hc complex, focused refined on transmembrane region
Deposition Authors: Yan RH ,
Li YN
,
Li YN  ,
Zhang YY
,
Zhang YY 
Sample: cryo EM map of Erastin-bound xCT-4F2hc complex, focused refined on transmembrane region
Deposition Authors: Yan RH
 ,
Li YN
,
Li YN  ,
Zhang YY
,
Zhang YY 
Structural basis for the recognition of SARS-CoV-2 by full-length human ACE2.
Abstract:
Angiotensin-converting enzyme 2 (ACE2) is the cellular receptor for severe acute respiratory syndrome-coronavirus (SARS-CoV) and the new coronavirus (SARS-CoV-2) that is causing the serious coronavirus disease 2019 (COVID-19) epidemic. Here, we present cryo-electron microscopy structures of full-length human ACE2 in the presence of the neutral amino acid transporter B0AT1 with or without the receptor binding domain (RBD) of the surface spike glycoprotein (S protein) of SARS-CoV-2, both at an overall resolution of 2.9 angstroms, with a local resolution of 3.5 angstroms at the ACE2-RBD interface. The ACE2-B0AT1 complex is assembled as a dimer of heterodimers, with the collectrin-like domain of ACE2 mediating homodimerization. The RBD is recognized by the extracellular peptidase domain of ACE2 mainly through polar residues. These findings provide important insights into the molecular basis for coronavirus recognition and infection.
Angiotensin-converting enzyme 2 (ACE2) is the cellular receptor for severe acute respiratory syndrome-coronavirus (SARS-CoV) and the new coronavirus (SARS-CoV-2) that is causing the serious coronavirus disease 2019 (COVID-19) epidemic. Here, we present cryo-electron microscopy structures of full-length human ACE2 in the presence of the neutral amino acid transporter B0AT1 with or without the receptor binding domain (RBD) of the surface spike glycoprotein (S protein) of SARS-CoV-2, both at an overall resolution of 2.9 angstroms, with a local resolution of 3.5 angstroms at the ACE2-RBD interface. The ACE2-B0AT1 complex is assembled as a dimer of heterodimers, with the collectrin-like domain of ACE2 mediating homodimerization. The RBD is recognized by the extracellular peptidase domain of ACE2 mainly through polar residues. These findings provide important insights into the molecular basis for coronavirus recognition and infection.
