EMD-35990
Cryo-EM structure of Mycobacterium tuberculosis OppABCD in the pre-translocation state
EMD-35990
Single-particle3.25 Å
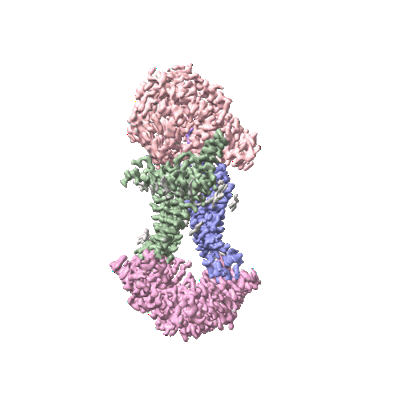 Deposition: 24/04/2023
Deposition: 24/04/2023Map released: 03/04/2024
Last modified: 31/07/2024
Sample Organism:
Mycobacterium tuberculosis (strain ATCC 25618 / H37Rv),
Mycolicibacterium smegmatis MC2 155
Sample: Cryo-EM structure of Mycobacterium tuberculosis OppABCD in the pre-translocation state
Fitted models: 8j5q (Avg. Q-score: 0.519)
Deposition Authors: Yang X ,
Hu T,
Zhang B
,
Hu T,
Zhang B  ,
Rao Z
,
Rao Z 
Sample: Cryo-EM structure of Mycobacterium tuberculosis OppABCD in the pre-translocation state
Fitted models: 8j5q (Avg. Q-score: 0.519)
Deposition Authors: Yang X
 ,
Hu T,
Zhang B
,
Hu T,
Zhang B  ,
Rao Z
,
Rao Z 
An oligopeptide permease, OppABCD, requires an iron-sulfur cluster domain for functionality.
Yang X  ,
Hu T,
Liang J,
Xiong Z,
Lin Z,
Zhao Y
,
Hu T,
Liang J,
Xiong Z,
Lin Z,
Zhao Y  ,
Zhou X,
Gao Y
,
Zhou X,
Gao Y  ,
Sun S,
Yang X
,
Sun S,
Yang X  ,
Guddat LW
,
Guddat LW  ,
Yang H
,
Yang H  ,
Rao Z
,
Rao Z  ,
Zhang B
,
Zhang B 
(2024) Nat Struct Mol Biol , 31 , 1072 - 1082
 ,
Hu T,
Liang J,
Xiong Z,
Lin Z,
Zhao Y
,
Hu T,
Liang J,
Xiong Z,
Lin Z,
Zhao Y  ,
Zhou X,
Gao Y
,
Zhou X,
Gao Y  ,
Sun S,
Yang X
,
Sun S,
Yang X  ,
Guddat LW
,
Guddat LW  ,
Yang H
,
Yang H  ,
Rao Z
,
Rao Z  ,
Zhang B
,
Zhang B 
(2024) Nat Struct Mol Biol , 31 , 1072 - 1082
Abstract:
Oligopeptide permease, OppABCD, belongs to the type I ABC transporter family. Its role is to import oligopeptides into bacteria for nutrient uptake and to modulate the host immune response. OppABCD consists of a cluster C substrate-binding protein (SBP), OppA, membrane-spanning OppB and OppC subunits, and an ATPase, OppD, that contains two nucleotide-binding domains (NBDs). Here, using cryo-electron microscopy, we determined the high-resolution structures of Mycobacterium tuberculosis OppABCD in the resting state, oligopeptide-bound pre-translocation state, AMPPNP-bound pre-catalytic intermediate state and ATP-bound catalytic intermediate state. The structures show an assembly of a cluster C SBP with its ABC translocator and a functionally required [4Fe-4S] cluster-binding domain in OppD. Moreover, the ATP-bound OppABCD structure has an outward-occluded conformation, although no substrate was observed in the transmembrane cavity. Here, we reveal an oligopeptide recognition and translocation mechanism of OppABCD, which provides a perspective on how this and other type I ABC importers facilitate bulk substrate transfer across the lipid bilayer.
Oligopeptide permease, OppABCD, belongs to the type I ABC transporter family. Its role is to import oligopeptides into bacteria for nutrient uptake and to modulate the host immune response. OppABCD consists of a cluster C substrate-binding protein (SBP), OppA, membrane-spanning OppB and OppC subunits, and an ATPase, OppD, that contains two nucleotide-binding domains (NBDs). Here, using cryo-electron microscopy, we determined the high-resolution structures of Mycobacterium tuberculosis OppABCD in the resting state, oligopeptide-bound pre-translocation state, AMPPNP-bound pre-catalytic intermediate state and ATP-bound catalytic intermediate state. The structures show an assembly of a cluster C SBP with its ABC translocator and a functionally required [4Fe-4S] cluster-binding domain in OppD. Moreover, the ATP-bound OppABCD structure has an outward-occluded conformation, although no substrate was observed in the transmembrane cavity. Here, we reveal an oligopeptide recognition and translocation mechanism of OppABCD, which provides a perspective on how this and other type I ABC importers facilitate bulk substrate transfer across the lipid bilayer.
