EMD-5443
Regulation of mammalian transcription by Gdown1 through a novel steric crosstalk revealed by cryo-EM
EMD-5443
Single-particle19.0 Å
 Deposition: 29/06/2012
Deposition: 29/06/2012Map released: 13/02/2013
Last modified: 13/02/2013
Sample Organism:
Bos taurus
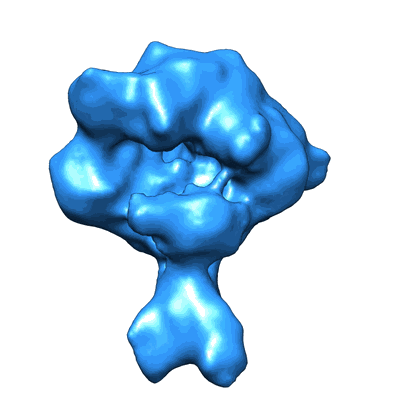
Sample: Bovine RNA polymerase II elongation complex with Gdown1
Deposition Authors: Wu YM, Chang JW, Wang CH ,
Lin YC,
Wu PL,
Huang SH
,
Lin YC,
Wu PL,
Huang SH  ,
Chang CC,
Hu X,
Gnatt A,
Chang WH
,
Chang CC,
Hu X,
Gnatt A,
Chang WH
Sample: Bovine RNA polymerase II elongation complex with Gdown1
Deposition Authors: Wu YM, Chang JW, Wang CH
 ,
Lin YC,
Wu PL,
Huang SH
,
Lin YC,
Wu PL,
Huang SH  ,
Chang CC,
Hu X,
Gnatt A,
Chang WH
,
Chang CC,
Hu X,
Gnatt A,
Chang WH
Regulation of mammalian transcription by Gdown1 through a novel steric crosstalk revealed by cryo-EM.
Wu YM,
Chang JW,
Wang CH  ,
Lin YC,
Wu PL,
Huang SH
,
Lin YC,
Wu PL,
Huang SH  ,
Chang CC,
Hu X,
Gnatt A,
Chang WH
,
Chang CC,
Hu X,
Gnatt A,
Chang WH
(2012) Embo J. , 31 , 3575 - 3587
 ,
Lin YC,
Wu PL,
Huang SH
,
Lin YC,
Wu PL,
Huang SH  ,
Chang CC,
Hu X,
Gnatt A,
Chang WH
,
Chang CC,
Hu X,
Gnatt A,
Chang WH
(2012) Embo J. , 31 , 3575 - 3587
Abstract:
In mammals, a distinct RNA polymerase II form, RNAPII(G) contains a novel subunit Gdown1 (encoded by POLR2M), which represses gene activation, only to be reversed by the multisubunit Mediator co-activator. Here, we employed single-particle cryo-electron microscopy (cryo-EM) to disclose the architectures of RNAPII(G), RNAPII and RNAPII in complex with the transcription initiation factor TFIIF, all to ~19 Å. Difference analysis mapped Gdown1 mostly to the RNAPII Rpb5 shelf-Rpb1 jaw, supported by antibody labelling experiments. These structural features correlate with the moderate increase in the efficiency of RNA chain elongation by RNAP II(G). In addition, our updated RNAPII-TFIIF map showed that TFIIF tethers multiple regions surrounding the DNA-binding cleft, in agreement with cross-linking and biochemical mapping. Gdown1's binding sites overlap extensively with those of TFIIF, with Gdown1 sterically excluding TFIIF from RNAPII, herein demonstrated by competition assays using size exclusion chromatography. In summary, our work establishes a structural basis for Gdown1 impeding initiation at promoters, by obstruction of TFIIF, accounting for an additional dependent role of Mediator in activated transcription.
In mammals, a distinct RNA polymerase II form, RNAPII(G) contains a novel subunit Gdown1 (encoded by POLR2M), which represses gene activation, only to be reversed by the multisubunit Mediator co-activator. Here, we employed single-particle cryo-electron microscopy (cryo-EM) to disclose the architectures of RNAPII(G), RNAPII and RNAPII in complex with the transcription initiation factor TFIIF, all to ~19 Å. Difference analysis mapped Gdown1 mostly to the RNAPII Rpb5 shelf-Rpb1 jaw, supported by antibody labelling experiments. These structural features correlate with the moderate increase in the efficiency of RNA chain elongation by RNAP II(G). In addition, our updated RNAPII-TFIIF map showed that TFIIF tethers multiple regions surrounding the DNA-binding cleft, in agreement with cross-linking and biochemical mapping. Gdown1's binding sites overlap extensively with those of TFIIF, with Gdown1 sterically excluding TFIIF from RNAPII, herein demonstrated by competition assays using size exclusion chromatography. In summary, our work establishes a structural basis for Gdown1 impeding initiation at promoters, by obstruction of TFIIF, accounting for an additional dependent role of Mediator in activated transcription.
