Function and Biology Details
Reaction catalysed:
Preferential cleavage: Arg-|-, Lys-|-.
Biochemical function:
Biological process:
Cellular component:
- not assigned
Sequence domains:
Structure domain:
Structure analysis Details
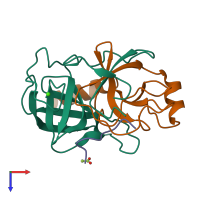
Assembly composition:
hetero trimer (preferred)
Assembly name:
Trypsin (preferred)
PDBe Complex ID:
PDB-CPX-133377 (preferred)
Entry contents:
3 distinct polypeptide molecules
Macromolecules (3 distinct):