Function and Biology Details
Reaction catalysed:
Thiol-dependent hydrolysis of ester, thioester, amide, peptide and isopeptide bonds formed by the C-terminal Gly of ubiquitin (a 76-residue protein attached to proteins as an intracellular targeting signal).
Biochemical function:
Biological process:
Cellular component:
- not assigned
Sequence domains:
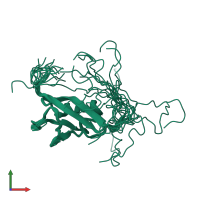
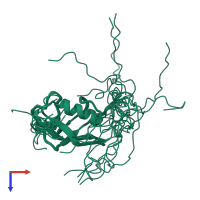
Structure domain:
Structure analysis Details
Assembly composition:
monomeric (preferred)
Assembly name:
Ubiquitin carboxyl-terminal hydrolase 14 (preferred)
PDBe Complex ID:
PDB-CPX-191649 (preferred)
Entry contents:
1 distinct polypeptide molecule
Macromolecule:
Ligands and Environments
No bound ligands
No modified residues
Experiments and Validation Details
wwPDB Validation report is not available for this NMR entry.
Refinement method:
torsion angle dynamics
Expression system: Not provided