Function and Biology Details
Reaction catalysed:
Limited hydrolysis of proteins of the neuroexocytosis apparatus, synaptobrevin (also known as neuronal vesicle-associated membrane protein, VAMP), synaptosome-associated protein of 25 kDa (SNAP25) or syntaxin. No detected action on small molecule substrates.
Biochemical function:
Biological process:
Cellular component:
- not assigned
Sequence domain:
Structure analysis Details
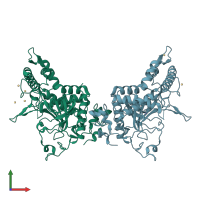
Assembly composition:
monomeric (preferred)
Assembly name:
Botulinum neurotoxin F light chain (preferred)
PDBe Complex ID:
PDB-CPX-151870 (preferred)
Entry contents:
1 distinct polypeptide molecule
Macromolecule: