Function and Biology Details
Reactions catalysed:
Exonucleolytic cleavage in the 3'- to 5'-direction to yield nucleoside 5'-phosphates
The C-O-P bond 3' to the apurinic or apyrimidinic site in DNA is broken by a beta-elimination reaction, leaving a 3'-terminal unsaturated sugar and a product with a terminal 5'-phosphate
Biochemical function:
Biological process:
Cellular component:
- not assigned
Structure analysis Details
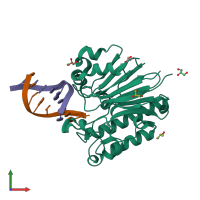
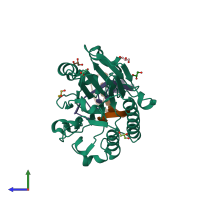
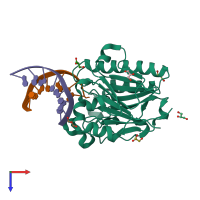
Assembly composition:
hetero trimer (preferred)
Assembly name:
DNA uridine endonuclease and DNA (preferred)
PDBe Complex ID:
PDB-CPX-116265 (preferred)
Entry contents:
1 distinct polypeptide molecule
2 distinct DNA molecules
2 distinct DNA molecules
Macromolecules (3 distinct):