Aldoxime dehydratase
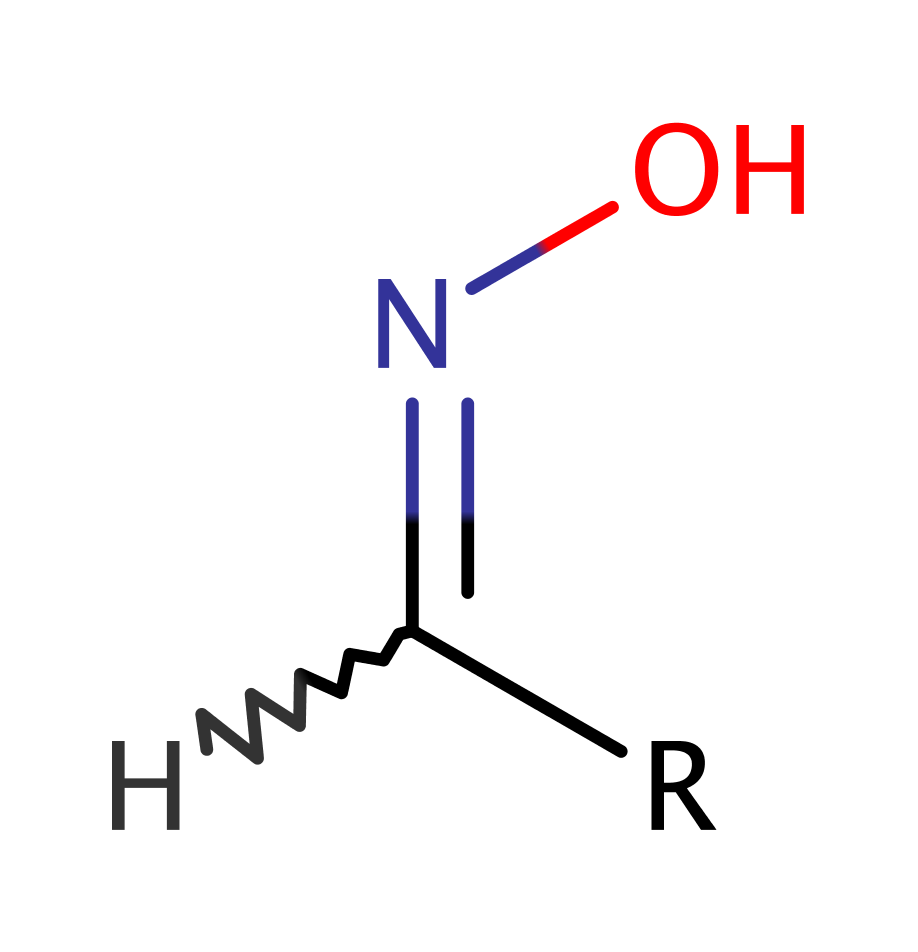
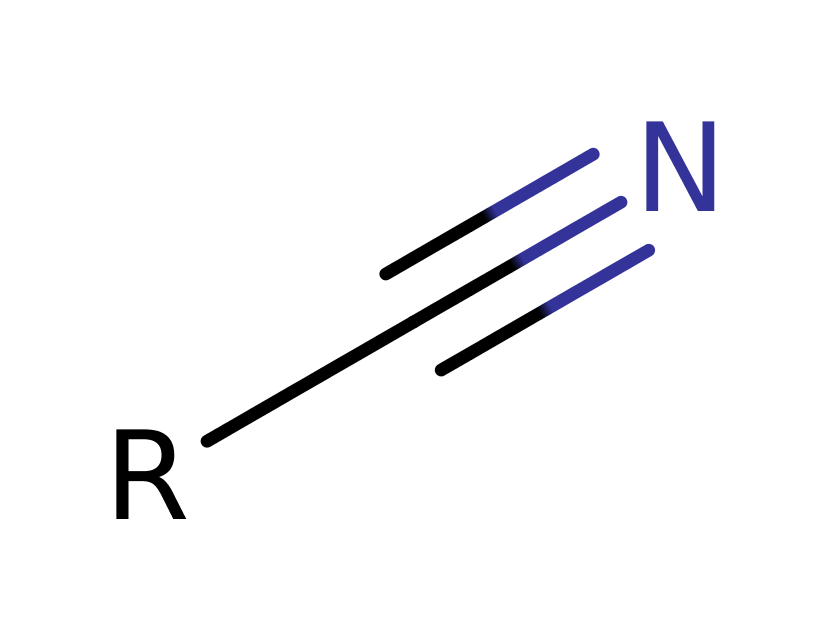
Aldoxime dehydratase (Oxd) is a heme-containing enzyme that catalyses the dehydration of aldoximes (R–CHN–OH) to the corresponding nitrile (R–C'N). Some species of microbes utilise an aldoxime-nitrile pathway, metabolising aldoximes to the corresponding carboxylic acids through nitriles formed by dehydration of aldoximes with aldoxime dehydratase. There are two pathways for the conversion of nitriles to carboxylic acids, i) the hydrolysis of nitriles by nitrilase, and ii) the sequential actions of nitrile hydratase and amidase. Nitriles are important for biological self-defence mechanisms, and are also of interest as intermediates in some industrial processes.
The dehydration reaction proceeds only via N-coordinated substrate in the ferrous heme. The organic substrate is shown by crystallographic and spectroscopic data to be directly coordinated to the heme iron in dehydration of aldoxime, which is a unique example among heme enzymes
Reference Protein and Structure
- Sequence
-
Q76K71

 (Sequence Homologues)
(PDB Homologues)
(Sequence Homologues)
(PDB Homologues)
- Biological species
-
Rhodococcus erythropolis (Bacteria)

- PDB
-
3a15
- Crystal Structure of Substrate-Free Form of Aldoxime Dehydratase (OxdRE)
(1.79 Å)



- Catalytic CATH Domains
- (see all for 3a15)
- Cofactors
- Heme b (1)
Enzyme Mechanism
Introduction
It is proposed that the dehydration reaction of the heme-bound aldoxime proceeds in a general acid-base catalysis with His320 working as a catalytic residue in the distal heme pocket. The residue His299 is coordinated as a distal ligand to the Fe(II) centre of the heme prosthetic group, directing the reaction site towards a non-polar region of the active site pocket, while several other residues are implicated in orientating the catalytic acid/base for optimum reactivity.
Catalytic Residues Roles
| UniProt | PDB* (3a15) | ||
| Phe306 | Phe306(326)A | Phe306 thought to be important for mediating the ratio of hydrophobic and hydrophilic character in the active site, which in turn controls the orientation of the haem and therefore the efficiency of reaction. | steric role |
| His320 | His320(340)A | Acts as a general acid/base. Correct orientation of His320 known to be crucial for catalytic activity. | hydrogen bond acceptor, hydrogen bond donor, proton acceptor, proton donor, activator, promote heterolysis |
| His299 | His299(319)A | heme axial ligand via tele nitrogen. | metal ligand, steric role |
| Glu143, Arg178 | Glu143(163)A, Arg178(198)A | Form a hydrogen bonding network with HIs320, holding this residue in the correct orientation and activating it to act as a general acid/base. | hydrogen bond acceptor, steric role |
| Ser219 | Ser219(239)A | Binds the substrate holding it in the correct orientation for the reaction to occur. | hydrogen bond acceptor, steric role |
Chemical Components
intramolecular elimination, proton transfer, radical formation, electron transfer, overall reactant used, cofactor used, overall product formed, native state of enzyme regenerated, native state of cofactor regeneratedReferences
- Sawai H et al. (2009), J Biol Chem, 284, 32089-32096. X-ray crystal structure of michaelis complex of aldoxime dehydratase. DOI:10.1074/jbc.M109.018762. PMID:19740758.
- Betke T et al. (2018), Chembiochem, 19, 768-779. Biocatalytic Synthesis of Nitriles through Dehydration of Aldoximes: The Substrate Scope of Aldoxime Dehydratases. DOI:10.1002/cbic.201700571. PMID:29333684.
- Nomura J et al. (2013), Proc Natl Acad Sci U S A, 110, 2810-2815. Crystal structure of aldoxime dehydratase and its catalytic mechanism involved in carbon-nitrogen triple-bond synthesis. DOI:10.1073/pnas.1200338110. PMID:23382199.
- Pan XL et al. (2012), J Phys Chem B, 116, 5689-5693. QM/MM study on the catalytic mechanism of heme-containing aliphatic aldoxime dehydratase. DOI:10.1021/jp302114d. PMID:22554192.
- Sato H et al. (2010), J Biol Chem, 285, 34793-34802. Novel isonitrile hydratase involved in isonitrile metabolism. DOI:10.1074/jbc.M110.150227. PMID:20826798.
- Konishi K et al. (2006), Proc Natl Acad Sci U S A, 103, 564-568. Discovery of a reaction intermediate of aliphatic aldoxime dehydratase involving heme as an active center. DOI:10.1073/pnas.0505412103. PMID:16407114.

Step 1. The Fe(II) centre donates a single electron to the N of the aldoxime, initiating the elimination of water, with concomitant proton abstraction from His320. OxdA reaction is an unusual example of a heme directly activating an organic substrate, unlike the utilization of peroxide or oxygen as a mediator of the catalysis, used by other heme-containing enzymes like P450.
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| Phe306(326)A | steric role |
| Arg178(198)A | hydrogen bond donor, steric role |
| Ser219(239)A | hydrogen bond acceptor, steric role |
| Glu143(163)A | hydrogen bond acceptor, steric role |
| His299(319)A | metal ligand, steric role |
| His320(340)A | activator, promote heterolysis, hydrogen bond donor, proton donor |
Chemical Components
ingold: intramolecular elimination, proton transfer, radical formation, electron transfer, overall reactant used, cofactor used, overall product formed
Step 2. The Fe(III) centre donates a second single electron to the N of the aldoxime. Spectroscopic studies suggest the Fe metal to adopt an oxidation state like that observed in horse radish peroxidase: a highly oxidised (+4) feryl-oxo heme species [PMID20826798:].
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| Phe306(326)A | steric role |
| Arg178(198)A | hydrogen bond donor, steric role |
| Ser219(239)A | hydrogen bond acceptor, steric role |
| Glu143(163)A | hydrogen bond acceptor, steric role |
| His299(319)A | metal ligand, steric role |
| His320(340)A | hydrogen bond acceptor |
Chemical Components
electron transfer, cofactor used
Step 3. His320 abstracts the imine alpha proton, forming a carbon, nitrogen triple bond. The Fe(iV) centre receives two electrons, regenerating the metal centre.
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| Phe306(326)A | steric role |
| Arg178(198)A | hydrogen bond donor, steric role |
| Ser219(239)A | hydrogen bond acceptor, steric role |
| Glu143(163)A | hydrogen bond acceptor, steric role |
| His299(319)A | metal ligand, steric role |
| His320(340)A | activator, hydrogen bond acceptor, proton acceptor |

 Download:
Download: