Vanadium-dependent bromoperoxidase (organic sulfide oxidation)
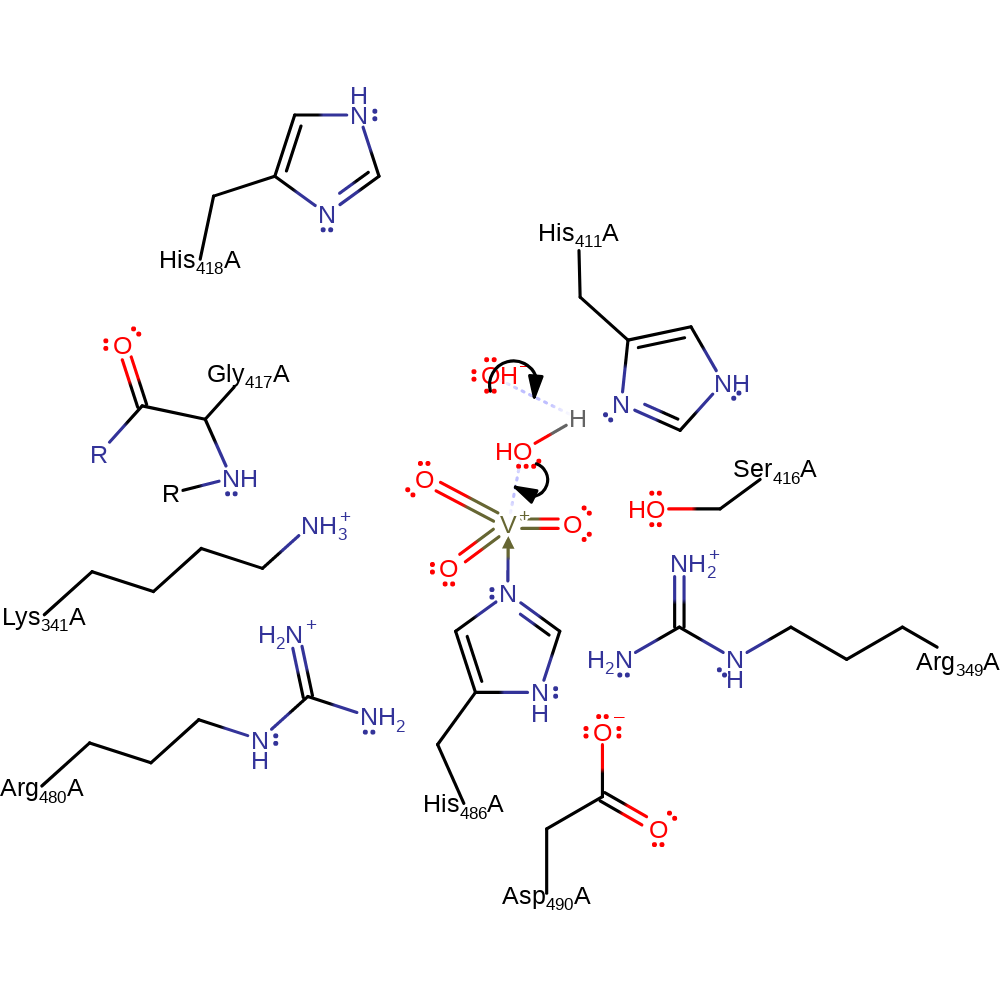
V-BPO, isolated from Ascophyllum nodosum, is a vanadium-dependent haloperoxidase. It catalyses the oxidation of halides by hydrogen peroxide to form hypohalides, which can go on to react with organic substrates in halogenation reactions. In this entry, we show one of the side reactions of the vanadium-dependent haloperoxidases: the oxidation or organic sulfides. V-BPO has the ability to oxidise bromide and, to a lesser extent, chloride and iodide as well as chlorinate organic hydrocarbons. V-BPO contains a vanadium(V) ion with a trigonal bipyramidal coordination sphere. It is coordinated to His486 and a hydroxide in the axial positions, and two oxygen atoms and a hydroxide in the equatorial positions.
Catalyzes the halogenation of organic substrates in the presence of hydrogen peroxide. This entry represents the enzyme's reaction with organic sulfides.
Reference Protein and Structure
- Sequence
-
P81701
 (1.11.1.18)
(1.11.1.18)
 (Sequence Homologues)
(PDB Homologues)
(Sequence Homologues)
(PDB Homologues)
- Biological species
-
Ascophyllum nodosum (Knotted wrack)

- PDB
-
1qi9
- X-RAY SIRAS STRUCTURE DETERMINATION OF A VANADIUM-DEPENDENT HALOPEROXIDASE FROM ASCOPHYLLUM NODOSUM AT 2.0 A RESOLUTION
(2.05 Å)



- Catalytic CATH Domains
-
1.10.606.10
 (see all for 1qi9)
(see all for 1qi9)
- Cofactors
- Vanadate(3-) (1)
Enzyme Reaction (EC:1.11.1.18)
Enzyme Mechanism
Introduction
An oxygen of hydrogen peroxide attacks the vanadium centre and causes the elimination of the axial hydroxide, which then deprotonates the bound peroxide to form water. The second oxygen of the peroxide ligand then attacks the vanadium centre and causes the elimination of an equatorial oxygen, which then abstracts the second peroxide proton. This forms a side-on vanadium-peroxo species. The sulfide attacks one of the oxygens of the peroxo group, causing the O-O bond to break and leading to the formation of an axial vanadium-O-sulfide complex. In the final step, the sulfate is eliminated and in an inferred return state a hydroxide deprotonates the histidine activated water to regenerate the cofactor.
Catalytic Residues Roles
| UniProt | PDB* (1qi9) | ||
| Lys341 | Lys341A | Lys341 is thought to polarise the vanadium-bound peroxo group and increase its susceptibility to attack by the sulfide. Lys341 forms a hydrogen bond to His411 and this is thought to modulate its polarising power. | electrostatic stabiliser |
| His411 | His411A | His411 forms a hydrogen bond to Lys341, buffering its polarising power towards the peroxo group. | increase basicity, electrostatic stabiliser, increase acidity |
| His418 | His418A | His418 increases the nucleophilicity of the axial hydroxide, thus making it more able to abstract a proton from hydrogen peroxide | electrostatic stabiliser |
| Arg349, Gly417 (main-N), Ser416, Arg480 | Arg349A, Gly417A (main-N), Ser416A, Arg480A | Forms the positive binding site for the cofactor; activating and stabilising it. | electrostatic stabiliser |
| His486 | His486A | His486 coordinates to the vanadate ion and is thought to be necessary for the formation of the vanadium-peroxo intermediate. It is thought that it is responsible for the lengthening and possible weakening of the axial V-O bond. | activator, metal ligand |
| Asp490 | Asp490A | Helps activate and position the vanadate binding His486. | activator |
Chemical Components
proton transfer, bimolecular nucleophilic addition, dehydration, acidic bimolecular nucleophilic substitution, intramolecular nucleophilic substitution, bimolecular nucleophilic substitution, unimolecular elimination by the conjugate base, inferred reaction stepReferences
- Dembitsky VM (2003), Tetrahedron, 59, 4701-4720. Oxidation, epoxidation and sulfoxidation reactions catalysed by haloperoxidases. DOI:10.1016/s0040-4020(03)00701-4.
- Waller MP et al. (2008), J Phys Chem B, 112, 5813-5823. 51V NMR Chemical Shifts from Quantum-Mechanical/Molecular-Mechanical Models of Vanadium Bromoperoxidase. DOI:10.1021/jp800580n. PMID:18412416.
- Zampella G et al. (2005), J Am Chem Soc, 127, 953-960. Reactivity of Peroxo Forms of the Vanadium Haloperoxidase Cofactor. A DFT Investigation. DOI:10.1021/ja046016x. PMID:15656634.
- ten Brink HB et al. (2001), Eur J Biochem, 268, 132-138. Sulfoxidation mechanism of vanadium bromoperoxidase fromAscophyllum nodosum. DOI:10.1046/j.1432-1327.2001.01856.x.
- Rehder D et al. (2000), J Inorg Biochem, 80, 115-121. Water and bromide in the active center of vanadate-dependent haloperoxidases. DOI:10.1016/s0162-0134(00)00047-7. PMID:10885471.
- Littlechild J (1999), Curr Opin Chem Biol, 3, 28-34. Haloperoxidases and their role in biotransformation reactions. DOI:10.1016/s1367-5931(99)80006-4.
- Weyand M et al. (1999), J Mol Biol, 293, 595-611. X-ray structure determination of a vanadium-dependent haloperoxidase from Ascophyllum nodosum at 2.0 Å resolution. DOI:10.1006/jmbi.1999.3179. PMID:10543953.
- Andersson M et al. (1997), J Org Chem, 62, 8455-8458. Asymmetric Sulfoxidation Catalyzed by a Vanadium-Containing Bromoperoxidase. DOI:10.1021/jo9712456.

Step 1. The axial hydroxide of the vanadate cofactor deprotonates hydrogen peroxide.
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His411A | increase basicity |
| His486A | metal ligand |
| Asp490A | activator |
| Lys341A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| His418A | electrostatic stabiliser |
| His486A | activator |
| Ser416A | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
Chemical Components
proton transfer
Step 2. The activated hydrogen peroxide initiates a nucleophilic attack on the vanadate in a substitution reaction, eliminating water.
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His486A | metal ligand |
| Asp490A | activator |
| His486A | activator |
| Lys341A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| His411A | electrostatic stabiliser |
| Ser416A | electrostatic stabiliser |
| Gly417A (main-N) | electrostatic stabiliser |
| His418A | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
Chemical Components
ingold: bimolecular nucleophilic addition, dehydration
Step 3. One of the equatorial oxo groups deprotonates the attached hydrogen peroxide.
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His486A | metal ligand |
| Asp490A | activator |
| His486A | activator |
| His418A | electrostatic stabiliser |
| Lys341A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| Ser416A | electrostatic stabiliser |
| Gly417A (main-N) | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
Chemical Components
ingold: acidic bimolecular nucleophilic substitution
Step 4. The peroxide initiates a nucleophilic attack on the vanadate in a substitution reaction, eliminating hydroxide and forming a three membered ring.
Download: Image, Marvin FileCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His486A | metal ligand |
| Asp490A | activator |
| His486A | activator |
| His411A | electrostatic stabiliser |
| Lys341A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| Ser416A | electrostatic stabiliser |
| Gly417A (main-N) | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
Chemical Components
ingold: intramolecular nucleophilic substitutionCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His486A | metal ligand |
| Asp490A | activator |
| His486A | activator |
| Lys341A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| Ser416A | electrostatic stabiliser |
| Gly417A (main-N) | electrostatic stabiliser |
| His418A | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
Chemical Components
ingold: bimolecular nucleophilic substitutionCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His486A | activator, metal ligand |
| Asp490A | activator |
| His418A | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
| Gly417A (main-N) | electrostatic stabiliser |
| Ser416A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| Lys341A | electrostatic stabiliser |
Chemical Components
ingold: unimolecular elimination by the conjugate baseCatalytic Residues Roles
| Residue | Roles |
|---|---|
| His486A | metal ligand |
| His411A | increase acidity |
| Lys341A | electrostatic stabiliser |
| Arg349A | electrostatic stabiliser |
| Ser416A | electrostatic stabiliser |
| Arg480A | electrostatic stabiliser |
| His486A | activator |
| Asp490A | activator |






 Download:
Download: