Viperin
Viperin (Virus Inhibitory Protein, ER-associated, Iterferon-inducible) is a member of the radical S-adenosyl-l-methionine (SAM) superfamily of enzymes, which typically employ a 4Fe-4S cluster to reductively cleave SAM to initiate chemistry. It has been shown that expression of viperin causes an increase in the fluidity of lipid membranes so as to impede the budding of nascent viral particles from the membrane which inhibits the propagation of the infection. It is now known that viperin catalyses the reductive addition of a 5'-deoxyadenosyl radical to UDP-glucose in vitro.
Reference Protein and Structure
- Sequence
-
Q8CBB9
 (4.2.-.-)
(4.2.-.-)
 (Sequence Homologues)
(PDB Homologues)
(Sequence Homologues)
(PDB Homologues)
- Biological species
-
Mus musculus (house mouse)

- PDB
-
5vsm
- Crystal structure of viperin with bound [4Fe-4S] cluster, 5'-deoxyadenosine, and L-methionine
(1.7 Å)



- Catalytic CATH Domains
- (see all for 5vsm)
- Cofactors
- Tetra-mu3-sulfido-tetrairon (1)
Enzyme Mechanism
Introduction
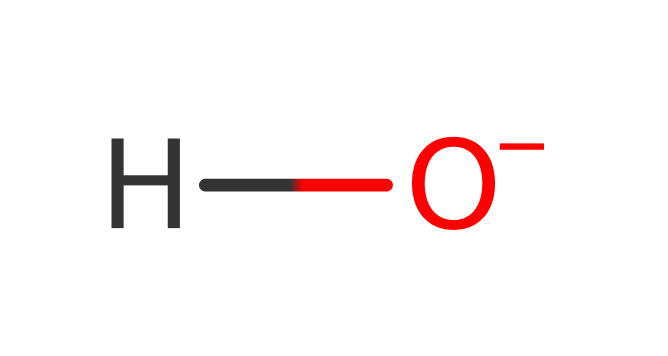
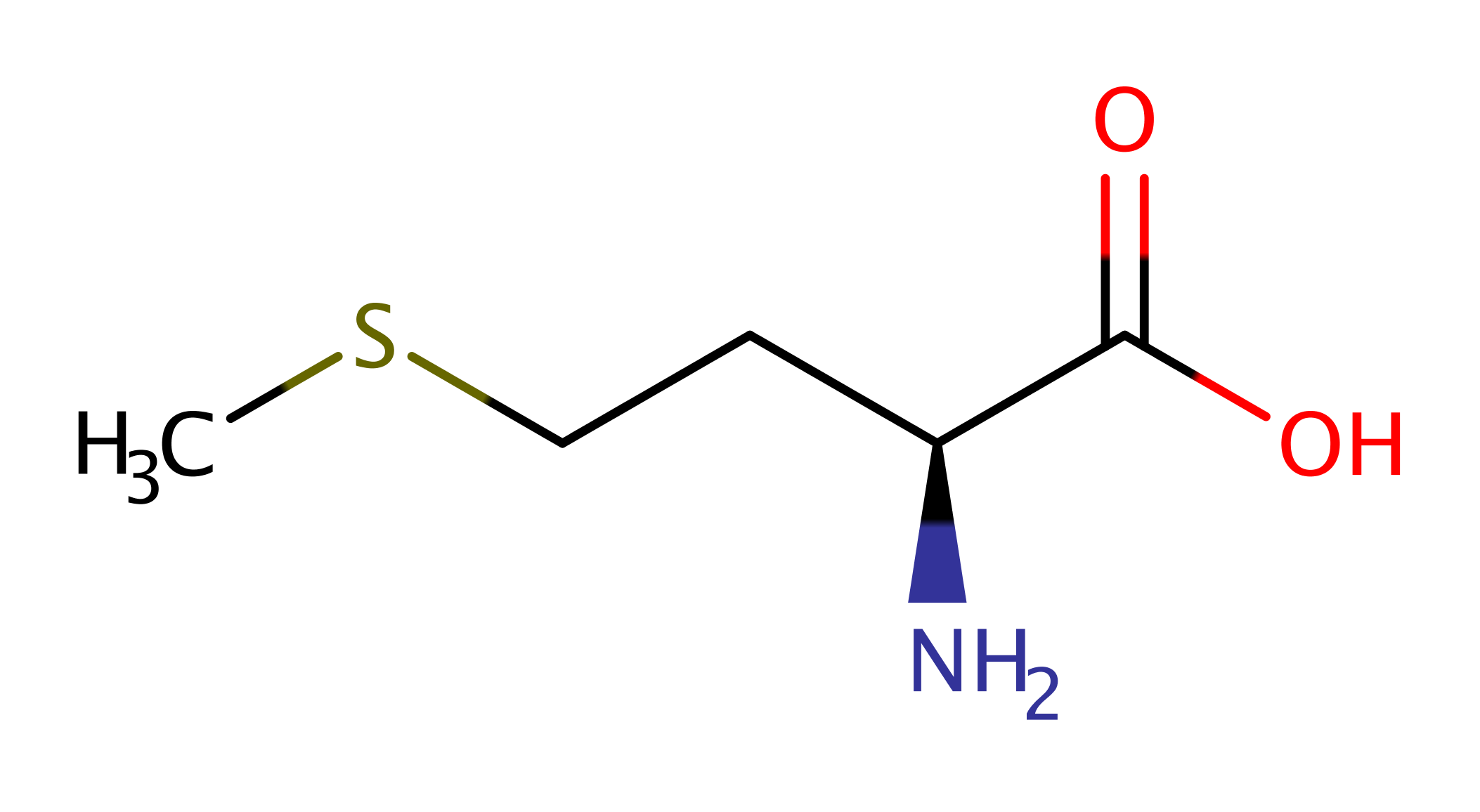
The exact product and hence mechanism of this enzyme is not yet known. The enzyme is known to catalyse the reductive addition of a 5'-deoxyadenosyl radical to UDP-glucose (in vitro). Performing the reaction in the presence of D2O shows that the hydrogen atom added to the UDP-glucose substrate comes from the solvent. Two moles of SAM are required for activity and the crystal structure shows that the enzyme likely activates the catalytic SAM using the canonical activation mechanism.
Catalytic Residues Roles
| UniProt | PDB* (5vsm) | ||
| Cys84, Cys88, Cys91 | Cys84(40)A, Cys88(44)A, Cys91(47)A | Forms the Iron-sulfur (4Fe-4S-S-AdoMet) binding site. | metal ligand |
Chemical Components
References
- Ebrahimi KH et al. (2017), FEBS Lett,The radical-SAM enzyme Viperin catalyzes reductive addition of a 5'-deoxyadenosyl radical to UDP-glucose in vitro. DOI:10.1002/1873-3468.12769. PMID:28752893.
- Fenwick MK et al. (2017), Proc Natl Acad Sci U S A, 114, 6806-6811. Structural studies of viperin, an antiviral radical SAM enzyme. DOI:10.1073/pnas.1705402114. PMID:28607080.
- Upadhyay AS et al. (2017), J Biol Chem,Cellular requirements for iron-sulfur cluster insertion into the antiviral radical SAM protein viperin. DOI:10.1074/jbc.M117.780122. PMID:28615450.
- Nelp MT et al. (2017), Biochemistry, 56, 3874-3876. Human Viperin Causes Radical SAM-Dependent Elongation of Escherichia coli, Hinting at Its Physiological Role. DOI:10.1021/acs.biochem.7b00608. PMID:28708394.
- Makins C et al. (2016), J Biol Chem, 291, 26806-26815. Does Viperin Function as a Radical S-Adenosyl-l-methionine-dependent Enzyme in Regulating Farnesylpyrophosphate Synthase Expression and Activity? DOI:10.1074/jbc.M116.751040. PMID:27834682.
- Helbig KJ et al. (2014), J Mol Biol, 426, 1210-1219. The role of viperin in the innate antiviral response. DOI:10.1016/j.jmb.2013.10.019. PMID:24157441.
- Haldar S et al. (2012), PLoS One, 7, e31797-. The presence of the iron-sulfur motif is important for the conformational stability of the antiviral protein, Viperin. DOI:10.1371/journal.pone.0031797. PMID:22363738.
- Duschene KS et al. (2010), FEBS Lett, 584, 1263-1267. The antiviral protein viperin is a radical SAM enzyme. DOI:10.1016/j.febslet.2010.02.041. PMID:20176015.
Catalytic Residues Roles
| Residue | Roles |
|---|---|
| Cys84(40)A | metal ligand |
| Cys88(44)A | metal ligand |
| Cys91(47)A | metal ligand |