EMD-30209
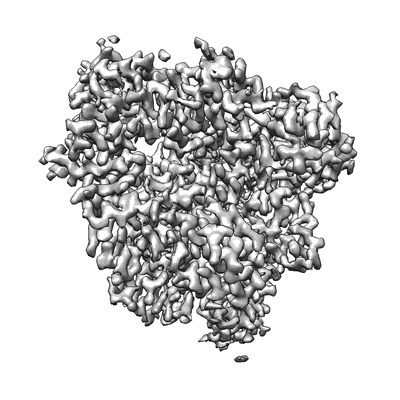
Cryo-EM structure of the apo nsp12-nsp7-nsp8 complex
EMD-30209
Single-particle2.8 Å
 Deposition: 09/04/2020
Deposition: 09/04/2020Map released: 22/04/2020
Last modified: 27/03/2024
Sample Organism:
Severe acute respiratory syndrome coronavirus 2,
SARS bat coronavirus
Sample: the apo nsp12-nsp7-nsp8 complex
Fitted models: 7bv1 (Avg. Q-score: 0.485)
Deposition Authors: Yin W ,
Mao C
,
Mao C 
Sample: the apo nsp12-nsp7-nsp8 complex
Fitted models: 7bv1 (Avg. Q-score: 0.485)
Deposition Authors: Yin W
 ,
Mao C
,
Mao C 
Structural basis for inhibition of the RNA-dependent RNA polymerase from SARS-CoV-2 by remdesivir.
Yin W  ,
Mao C
,
Mao C  ,
Luan X
,
Luan X  ,
Shen DD
,
Shen DD  ,
Shen Q
,
Shen Q  ,
Su H
,
Su H  ,
Wang X
,
Wang X  ,
Zhou F
,
Zhou F  ,
Zhao W
,
Zhao W  ,
Gao M
,
Gao M  ,
Chang S
,
Chang S  ,
Xie YC
,
Xie YC  ,
Tian G
,
Tian G  ,
Jiang HW,
Tao SC
,
Jiang HW,
Tao SC  ,
Shen J
,
Shen J  ,
Jiang Y
,
Jiang Y  ,
Jiang H
,
Jiang H  ,
Xu Y
,
Xu Y  ,
Zhang S
,
Zhang S  ,
Zhang Y
,
Zhang Y  ,
Xu HE
,
Xu HE 
(2020) Science , 368 , 1499 - 1504
 ,
Mao C
,
Mao C  ,
Luan X
,
Luan X  ,
Shen DD
,
Shen DD  ,
Shen Q
,
Shen Q  ,
Su H
,
Su H  ,
Wang X
,
Wang X  ,
Zhou F
,
Zhou F  ,
Zhao W
,
Zhao W  ,
Gao M
,
Gao M  ,
Chang S
,
Chang S  ,
Xie YC
,
Xie YC  ,
Tian G
,
Tian G  ,
Jiang HW,
Tao SC
,
Jiang HW,
Tao SC  ,
Shen J
,
Shen J  ,
Jiang Y
,
Jiang Y  ,
Jiang H
,
Jiang H  ,
Xu Y
,
Xu Y  ,
Zhang S
,
Zhang S  ,
Zhang Y
,
Zhang Y  ,
Xu HE
,
Xu HE 
(2020) Science , 368 , 1499 - 1504
Abstract:
The pandemic of coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has become a global crisis. Replication of SARS-CoV-2 requires the viral RNA-dependent RNA polymerase (RdRp) enzyme, a target of the antiviral drug remdesivir. Here we report the cryo-electron microscopy structure of the SARS-CoV-2 RdRp, both in the apo form at 2.8-angstrom resolution and in complex with a 50-base template-primer RNA and remdesivir at 2.5-angstrom resolution. The complex structure reveals that the partial double-stranded RNA template is inserted into the central channel of the RdRp, where remdesivir is covalently incorporated into the primer strand at the first replicated base pair, and terminates chain elongation. Our structures provide insights into the mechanism of viral RNA replication and a rational template for drug design to combat the viral infection.
The pandemic of coronavirus disease 2019 (COVID-19), caused by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), has become a global crisis. Replication of SARS-CoV-2 requires the viral RNA-dependent RNA polymerase (RdRp) enzyme, a target of the antiviral drug remdesivir. Here we report the cryo-electron microscopy structure of the SARS-CoV-2 RdRp, both in the apo form at 2.8-angstrom resolution and in complex with a 50-base template-primer RNA and remdesivir at 2.5-angstrom resolution. The complex structure reveals that the partial double-stranded RNA template is inserted into the central channel of the RdRp, where remdesivir is covalently incorporated into the primer strand at the first replicated base pair, and terminates chain elongation. Our structures provide insights into the mechanism of viral RNA replication and a rational template for drug design to combat the viral infection.
